|
8/18/2023 0 Comments Keynote 042 update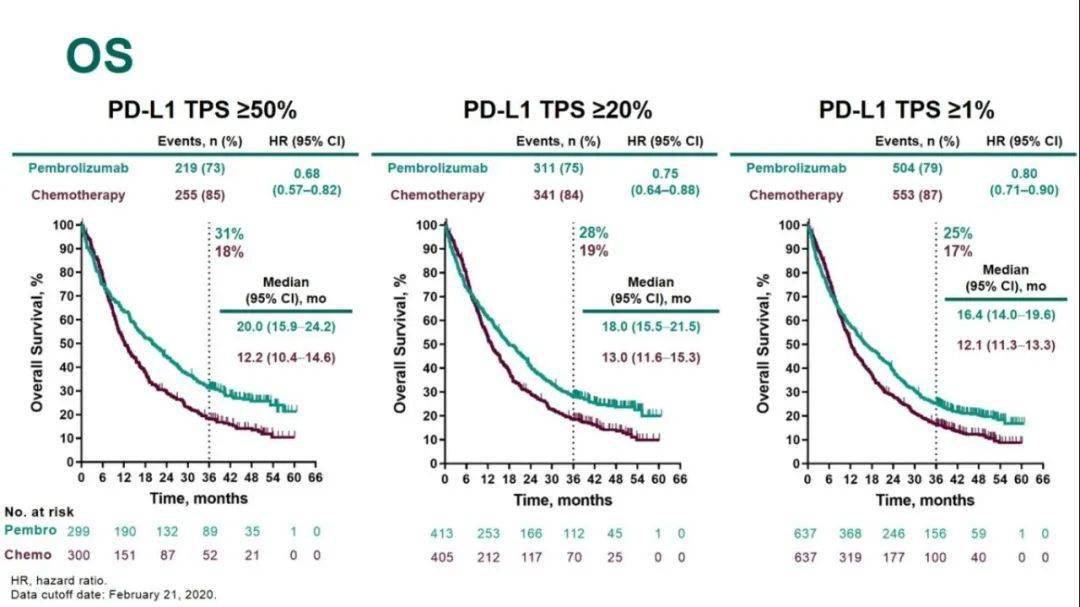
Treatment-related adverse events have long been a concerning issue of concurrent therapy. nonsquamous) is suggested in cohort A to further understand adverse event profile differences between the subgroups. Moreover, subgroup analysis according to histologic type (squamous vs. These are reminiscent of the typical adverse reactions to paclitaxel and carboplatin that were adopted by cohort A. Besides, infusion reactions of any grade occurred in 10 patients (8.9%) in cohort A and 2 (2.0%) in cohort B. 4.9%), neutrophil count decreased & neutropenia of grade 3-5 (25% vs. Patients in cohort A were more prone to hematological toxicities than those in cohort B, such as thrombocytopenia of any grade (16.1% vs. 70.6%).Īside from the adverse events leading to discontinuation of treatments and death that occurred more often in cohort A than B, some toxicities more observed in cohort A may be related to the chemotherapy regimen (Table 3). This study aimed to evaluate treatment outcomes and safety of the combination therapy with different platinum-doublet chemotherapies, which showed that both regimens were associated with comparable efficacy (ORR, 70.5% vs. 2021 Jun 4) is the largest trial to date of concurrent anti–PD-(L)1 therapy plus concurrent chemoradiation therapy (cCRT) in patients with previously untreated, locally advanced, stage III NSCLC. A recent study by the same group titled Pembrolizumab plus Concurrent Chemoradiation Therapy in Patients with Unresectable, Locally Advanced, Stage III Non–Small Cell Lung Cancer the Phase 2 KEYNOTE-799 Nonrandomized Trial (JAMA Oncol. To improve outcomes for patients with locally advanced non–small cell lung cancer who do not respond well to immunotherapy, efforts have been made at combining immunotherapy concurrently with chemoradiotherapy by Salma K. Shared Decision Making and Communication.Scientific Discovery and the Future of Medicine.Health Care Economics, Insurance, Payment.Clinical Implications of Basic Neuroscience. 
Challenges in Clinical Electrocardiography.However, single-agent pembrolizumab appears to be a feasible option, Mok concludes. Notably, the standard of care for patients with a TPS <50% is pembrolizumab plus chemotherapy. These data suggested that a lot of the benefit seen in the overall population was driven by patients with a TPS ≥50%, Mok says. Researchers also looked at the exploratory analysis of patients with a TPS <50%, where an initial OS improvement was not seen. A similar benefit was observed in patients with TPS ≥20% and ≥1%, Mok adds. Updated data for KEYNOTE-042 presented by Mok at the 2019 European Lung Cancer Congress showed a confirmed improvement in OS with pembrolizumab versus chemotherapy at 20 months versus 13 months in patients with a TPS ≥50%. Though, the primary endpoint in this trial was overall survival (OS) in patients with a tumor proportion score (TPS) ≥50%, ≥20%, and ≥1%. On the other hand, KEYNOTE-042 selected patients with PD-L1 ≥1%, and then did the same randomization of the PD-1 inhibitor versus chemotherapy. The difference between the 2 trials is that KEYNOTE-024 selected patients who had PD-L1 expression ≥50% and randomized them to receive either pembrolizumab or chemotherapy, using progression-free survival as the primary endpoint. The KEYNOTE-042 study is a sister study of KEYNOTE-024, Mok says. Mok, BMSc, MD, FRCPC, professor, Department of Clinical Oncology, Chinese University of Hong Kong, discusses updated data with pembrolizumab (Keytruda) in patients with non-small cell lung cancer (NSCLC).
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
